  
In February 2022, GSK halted vaccination in its phase 3 trials of its maternal RSV vaccine after finding an increased risk of preterm birth in vaccinated mothers, mainly in low and middle income countries. The European Medicines Agency is also set to make a decision about the vaccine later this year. Food and Drug Administration is expected by August. The vaccine has not yet been approved for use, but a decision by the U.S. In 2019, an estimated 3.6% of all deaths worldwide in children aged 1–6 months were due to RSV, with 97% of these deaths occurring in low and middle income countries. RSV is very common but can be fatal, especially in young children. Pfizer's maternal RSV vaccine aims to protect infants from severe illness caused by the respiratory syncytial virus (RSV). However, those who received the J&J/Janssen vaccine are now eligible for a booster shot if they received their initial dose at least 2 months ago.Pfizer says its vaccine is safe and effective, but experts contacted as part of an investigation published by The BMJ today, say Pfizer's trial data should be reviewed in light of the signal for preterm births seen in GSK's trial. At this time, the CDC is not recommending an additional dose for patients who received the Johnson & Johnson/Janssen vaccine, even if they are immunocompromised. This recommendation only applies to individuals who received an mRNA vaccine, in other words, the Pfizer or Moderna vaccine. Active treatment with immune-suppressing medications, including high-dose corticosteroids, chemotherapy, tumor necrosis factor (TNF) inhibitors, and other biologic agents that suppress or modulate the immune system.Moderate or severe primary immunodeficiency.Previous solid organ transplant (such as a kidney or liver transplant). 
Previous stem cell transplant or CAR T-cell therapy.This includes those with the following conditions: The CDC recommends a third dose for patients ages 5 and up who are moderately or severely immunocompromised. Read additional CDC guidance for the immunocompromised, including information for those receiving J&J vaccines and boosters. If you are between 5 and 11 years old, you are eligible for a Pfizer monovalent booster if you completed your 3-dose primary series of Pfizer at least 3 months ago. Those who are 18+ years old are eligible for a Pfizer or Moderna bivalent booster. Those who are between 12 and 17 years old are eligible for Pfizer bivalent booster only at this time. If you are immunocompromised, are 12 years of age or older, and have completed your primary series or your most recent booster at least 2 months ago, you are eligible for a bivalent booster.If you received the Johnson & Johnson/Janssen vaccine at least 2 months ago and are 18 years old or older, you are eligible to receive a bivalent booster shot.For people ages 5-11 years old, only the Pfizer monovalent booster is approved at this time. If you are 12 years or older and received the COVID-19 vaccine at least 2 months ago, you are eligible to receive a bivalent booster shot. 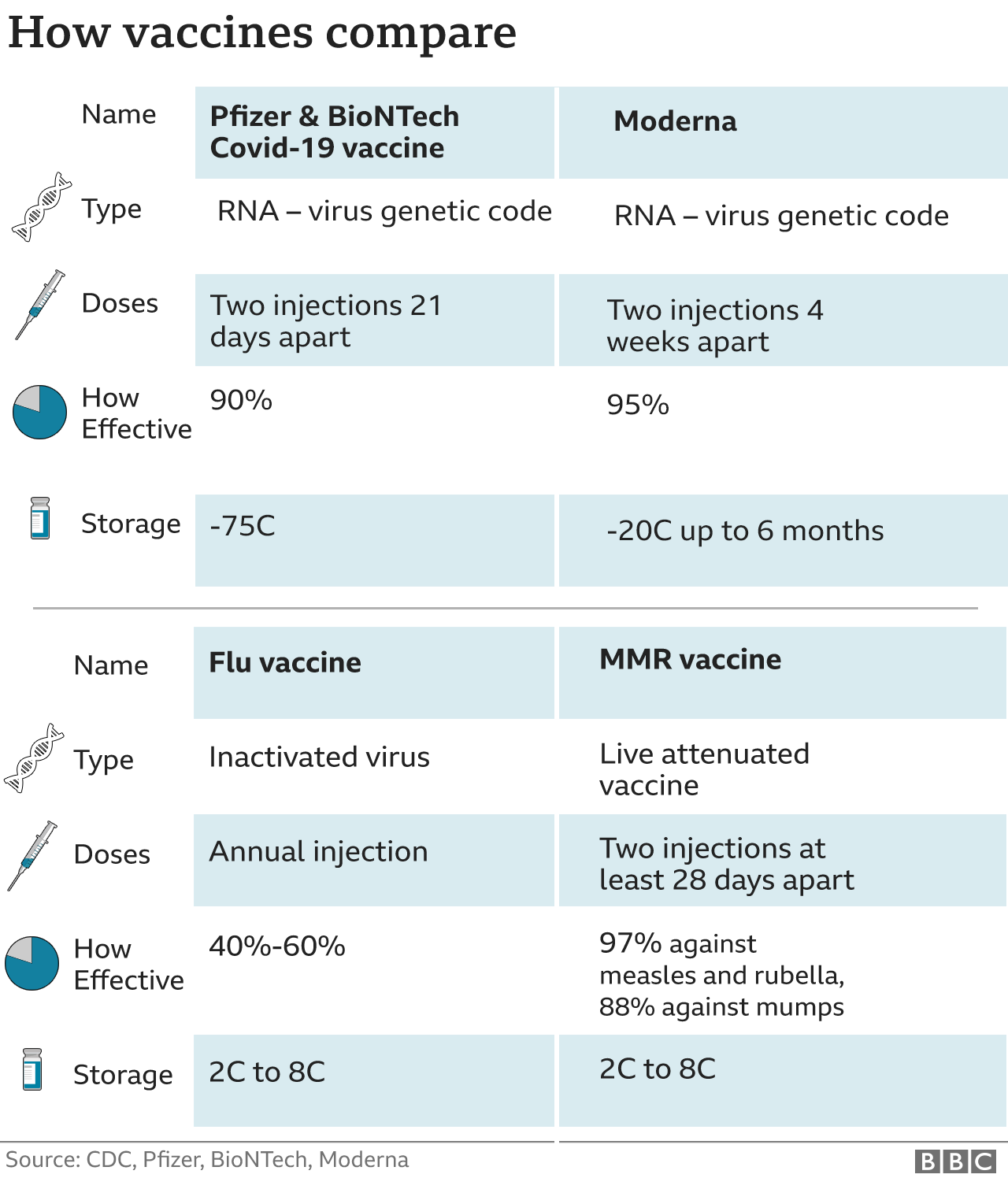
Please note, we will not take walk-ins for third doses.Įveryone ages 5 and older can get a booster shot. You can also speak with your health care provider about your medical condition and whether getting an additional dose is appropriate for you. If you are unsure if the medication you take is an immune-suppressing or immune-modulating medicine, which would make you eligible for a third dose of the vaccine, please see this list of medications (this list is not intended to be all-inclusive).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
